| VIVO Pathophysiology | Gastrointestinal Hormones |
Glucose-dependent Insulinotropic Peptide (Gastric Inhibitory Peptide)
This hormone was originally described as a peptide in intestinal extracts that inhibited acid secretion and motility in the stomach and was named "gastric inhibitory peptide". Subsequently, it was found that it had more potent effects by mimicking and enhancing secretion of insulin from the pancreas and is therefore now more commonly known as glucose-dependent insulinotropic peptide. Fortuituously, both of these names can be abbreviated as GIP.
To add a bit more confusion to hormone nomenclature, GIP, along with glucagon-like peptide-1 are known as "incretins". The incretin effect was coined many years ago to describe the observation that oral administration of glucose stimulates considerably higher levels of insulin release than if glucose is infused intravenously to achieve the same blood glucose concentration, suggesting that glucose in the gut induces secretion of something (the incretin) that potently enhances secretion of insulin from the pancreas.
GIP is a 40 amino acid peptide closely related to secretin. It is secreted from K cells, which are a type of endocrine cell in the epithelium of the small intestine. GIP is secreted into the circulation when K cells detect elevated concentrations of glucose, amino acids or log-chain fatty acids within the lumen of the small intestine.
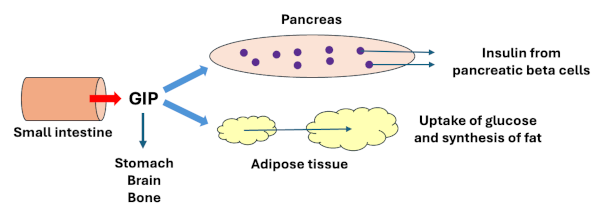
Ingestion of a meal results in secretion of insulin from pancreatic beta cells and it turns out that 60-80% of this insulin response is due to stimulation of the beta cells by GIP, rather than to direct effects of increased blood glucose concentrations on the beta cell. In turn, the rise in circulating insulin inhibits further secretion of GIP from K cells - a classical negative feedback loop.
GIP exerts its metabolic effects in two ways:
- GIP directly stimulates secretion of insulin: GIP binds to receptors on pancreatic beta cells and stimulates them to secrete insulin. In other words, GIP strongly enhances the glucose-dependent stimulation of insulin release.
- GIP acts indirectly to mimick the activity of insulin: Like insulin, GIP stimulates adipocytes to take up glucose and synthesize triglyceride, thereby increasing stores of energy as fat. This is referred to as an insulin-mimetic effect of GIP.
GIP has a number of other physiologic effects aside from its insulin and insulin-like effects:
- It inhibits secretion of acid in the stomach and suppresses gastric motility.
- In concert with several other factors, GIP acts in the brain and appears to have a role in regulating appetite and energy expenditure.
- GIP promotes proliferation of osteoblasts in bone, enhancing mineralization and promoting strengthening of bone.
As with any hormone, secretion of too little or too much GIP would be expected to have adverse consequences, and from the effects described above, you would be correct to deduce that aberrent secretion of GIP could have significant impacts on energy metabolism, obesity and hunger. Indeed, there is substantial evidence that GIP can be involved espectially in disease states including diabetes and obesity, and pharmacologic approaches to manipulating GIP and other incretins are being vigorously pursued.
References
Nauck MA, Meier JJ. Incretin hormones: Their role in health and disease. Diabetes Obes Metab. 2018; Suppl 1:5-21.
Rehfeld JF. The Origin and Understanding of the Incretin Concept. Front Endocrinol (Lausanne). 2018; 9:387.
Wolfe MM, Boylan MO, Chin WW. Glucose-Dependent Insulinotropic Polypeptide in Incretin Physiology: Role in Health and Disease. Endocr Rev. 2025; 46:479-500.
 Motilin Motilin |
Enteroglucagons  |
Updated March 2026. Send comments to Richard.Bowen@colostate.edu