| VIVO Infectious Diseases | Bacterial Diseases of Man and Animals |
Diagnostic Testing for Brucellosis
There are no pathognomonic clinical signs of brucellosis in either humans or animals, and application of laboratory diagnostic testing is critical to establish a diagnosis in any species. In humans, cases of brucellosis are often classified as suspected, probable, or confirmed. A diagnosis of probable is usually based on clinical observations and judgement, whereas a confirmed diagnosis requires laboratory testing. Accurate diagnosis allows proper treatment of confirmed cases and avoidance of unnecessary or ineffective treatment if brucellosis is not the problem.
There are three core methods that are applied to diagnosis of brucellosis:
- Isolation of the organism in culture
- Serologic testing to detect the presence of antibodies to Brucella
- Nucleic acid amplification and detection assays.
As described below, each of these techniques has strengths and weaknesses, and a combination of tests is often the most reliable. It is important to recognize that laboratory diagnosis of brucellosis is anything but straightforward, particular in regions where the disease is endemic.
Isolation of Brucella from Blood and Other Sources

Brucellosis seems to always involve a bacteremic stage, but the magnutide and duration of Brucella organisms in blood vary widely. Cultures of peripheral blood represent a suitable tool for confirming the disease, although a broad range (10 to 90%) in sensitivity for culture has been described in different reports. Early in the course of infection, low mangnitude but persistent bacteremia is usually seen and culture is often successful for isolation. However, as the disease becomes more chronic, the organism is taken up into macrophages and levels of bacteria in blood drop and become sporadic. Regardless, it is often important to culture 2 or 3 sequential blood samples to maximize probability of detecting the organism.
Frequently, bacteremic blood contains only 1 to 5 CFU per ml and culture of 20-30 ml is recommended for adults.
Blood cultures for brucellosis are commonly conducted using biphasic culture flasks (see image to right) that contain both broth and an agar medium slant (e.g. Castañeda flask) maintained in 10% CO2. Whole blood is inoculated into the broth and if Brucella is present it will grow in the broth. Every few days, the flask is tilted so that the agar slant is exposed to the broth, and if Brucella are present, they will form colonies on the agar. Such cultures should be maintained for at least 4 weeks.
The major disadvantages to culture-detection of Brucella and the time required for growth and biohazards associated with culture. Humans can be infected with as little as 10-100 CFU of Brucella, typically via the respiratory or conjunctival routes. It has been estimated that 2% of all human cases of brucellosis are the result of laboratory-based infection.
Serologic Testing for Antibodies to Brucella
Serological tests remain the most commonly used techniques for diagnosing brucellosis, especially in resource-poor settings. However, it is important to recognize that they have relatively poor specificity and results can be very difficult to interpret in individuals repeatedly exposed to Brucella organisms. It is also difficult to distinguish between active and past infections using serology because even IgM antibodies persist for long periods in many people.
The quest to develop diagnostically-reliable tests for brucellosis, particularly for ruminants, numerous serologic tests have been developed, some very simple and others complex. None of these tests are without fault and it is often recommended that a positive reaction with a given test be followed by confirmatory testing using a different test.
Rose Bengal Plate Agglutination Test (RBT)
The RBT is arguably the the most commonly-used and valuable serologic test for determining whether a human or animal has been, at some point, infected with Brucella melitensis, abortus or suis. This testing is conceptually and technical simple: Start with a standard suspension of phenol-killed Brucella (usually B. abortus that has been stained with rose bengal dye to give them a purple color. On a white plate or glass slide, mix a small volume of the stained bacteria with an equal volume of patient serum, incubate at room temperature with rocking for about 5 minutes, and examine for agglutination. An example of non-agglutinated (negative) and agglutinated (positive) samples are shown below.
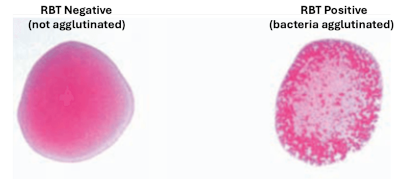
In some situations, dilutions of serum can be tested using RPT to provide an estimate of the magnitude of the antibody response. The RBT test is very high sensitivity and moderate specificity, again meaning that positive results should be confirmed using a different technique.
The is another test related to but distinct from RBT that is called a Febrile Antigen Agglutination Test or FBAT. This test is not recommended as it has quite poor sensitivity and specificity.
Serum Agglutination Test (SAT)
This was the original test reported by Bruce and colleagues in 1897, and is still considered a standard test today. It is conducted very similarly to the RBT but in tubes or microplates and by testing serial two-fold dilutions of serum (1:20 to 1:320) instead of just undiluted serum. The serum-bacteria mixtures are incubated overnight at 37C and then examined for agglutination. Agglutination is detected by seeing a clump of bacteria at the bottom of the tube, covered by a clear supernate; the mixture remains cloudy if agglutination has not occurred. The endpoint titer is the highest dilution of serum that results in visible agglutination. Like RBT, the SAT will detect both IgG and IgM antibodies against Brucella. A SAT titer of at least 80 is interpreted as a positive titer.
2-Mercaptoethanol Test (2-ME)
A common problem with agglutination tests is poor specificity due to crossreacting IgM antibodies. To destroy IgM but retain IgG antibodies, 2-mercaptoethnol (2-ME) or, if you dislike the smell of rotten eggs, dithiothreitol (DTT) is added to a standard serum agglutination test. IgG antibodies will be retrained but IgM antibodies destroyed by the 2-ME or DTT. It is often recommended to run SAT plus 2-ME tests in parallel.
Non-agglutinating Tests: ELISA
A large number of enzyme-linked immunosorbant assays are commercially available for serologic testing in humans or animals. The most common forms of such ELISA tests are indirect or competitive.
Other Types of Serologic Tests for Brucellosis
Several other tests have been developed and userd overy the years for diagnosis of brucellosis in man and animals, including complement fixation and fluorescence polarization tests. Also, simple point-of-care tests such as lateral flow or dipstick tests have some utility in envinroments that lack any laboratory facilities.
One other class of Brucella antibody tests that have been widely used in cattle are those to detect antibodies in milk samples. These tests are valuable because it is considerably easier to obtain milk samles from dairy cattle or buffalo than blood samples. The classical milk test is called a milk-ring test, but several companies market ELISA tests that have been validated for use with milk samples.
One final note of importance: the agglutination tests and some of ELISA tests only work for antibodies to rough strains of Brucella such as melitensis, abortus and suis. They will not, for example, detect antibodies to smooth strains like B. canis, for which different antigens are required.
Interpretation of Brucella Serologic Testing
As indicated at the beginning of this section, serologic testing is a valuable and very widely used technique for diagnosing brucellosis, but is far from free of complications. Signficant problems inclue:
- Significant variability among individuals in their antibody responses in both acute, chronic and recrudescent case of brucellosis.
- Cross-reactive immune responses to other bacteria.
- It is often very difficult to differentation active infection from past infections.
One standard recommendation for serologic diagnosis of recent infection with Brucella is finding a 4-fold increase in antibody titer (usually SAT or quantitative RBT) between acute and convalescent samples collected at lest 2 weeks apart.
Nucleic Acid Amplification Tests for Detection of Brucella
As with virtually all other infectious organisms, polymerase chain reaction (PCR) has become widely used for detecting Brucella DNA in samples from humans and other animals. The most frequently targeted gene to amplify is BCSP31.
For diagnosing brucellosis in animals, the most common samples tested are fluids or tissues from cases of abortion, which typically contain large numbers of organisms. PCR can be used in such cases to determine whether the abortion was indeed due to brucellosis or to other common causes of abortion such as Coxiella.
PCR is also widely used for diagnosis of human brucellosis. The question of what sample to use from suspected cases has been evaluated in several studies and the consensus appears to be that serum is better than whole blood. The problem with whole blood seems to be the inhibitor effect of heme from lysed red blood cells or high concentrations of human genomic DNA, both of which suppress PCR amplification of Brucella DNA, which is present in very low concentration. Brucella organisms are located predominantly within white blood cells, but apparently, its DNA is released into blood as bacteria break down or infected cells lyse, providing a target for DNA amplication.
One of the issues in using PCR for diagnosis of brucellosis in humans relates to its exquisite sensitivity, which can result in a positive test in patients that have been successfully treated due to minute amounts of non-viable organisms still circulating in blood. Such false positive tests for active infection may lead to unnecessary additional treatment.
In addition to conventional or real-time PCR, other DNA amplification techniques have been applied to diagnosing brucellosis such as loop-mediated isothermal amplification (LAMP), which is useful especially for laboratories that do not have a thermocycler.
References
Freire ML, Machado de Assis TS, Silva SN, Cota G. Diagnosis of human brucellosis: Systematic review and meta-analysis. PLoS Negl Trop Dis. 2024; 18:e0012030.
Qureshi KA, Parvez A, Fahmy NA, Abdel Hady BH, Kumar S, Ganguly A, Atiya A, Elhassan GO, Alfadly SO, Parkkila S, Aspatwar A. Brucellosis: epidemiology, pathogenesis, diagnosis and treatment-a comprehensive review. Ann Med. 2023; 55(2):2295398.
World Organization for Animal Health. Brucellosis
Yagupsky P, Morata P, Colmenero JD. Laboratory Diagnosis of Human Brucellosis. Clin Microbiol Rev. 2019; 33:e00073-19.
 Brucella species and Brucellosis: Introduction and Index Brucella species and Brucellosis: Introduction and Index |
Updated January 2026. Send comments to Richard Bowen: rbowen@colostate.edu