VIVO Pathophysiology
Atrial Naturetic Hormone
Atrial naturetic hormone or ANP is a member of a family of peptides that have important roles in regulating blood pressure, most prominently through their activity in the kidney to promote excretion of water and sodium. A majority of ANP is synthesized and secreted from cardiac muscle cells, particularly in the atria. Thus, the hormone was named for its source (atrial), major physiologic effect (naturesis or excretion of sodium), and its peptide nature. ANP can be thought of as an anti-hypertensive hormone and plays a central role in the regulation of blood pressure.
Structure, Synthesis, and Receptors

ANP is initially synthesized within cardiac myocytes as the prohormone preproANP. The biologically-active peptide ANP is release through a series of proteolytic cleavages and in humans is 28 amino acids in length that contains a loop created by a disulfide bond between two cysteines. Low levels of ANP are synthesized in a variety of other tissues and in certain pathologic states (e.g. heart failure), substantial synthesis occurs in the ventricles of the heart.
The naturetic peptide family includes several other peptides in addition to ANP:
- B-type naturetic peptide (BNP): originally identified in the brain, most of this peptide is synthesized in ventricular myocytes in the heart in response to cardiac stretch similarly to ANP.
- C-type naturetic peptide (CNP): synthesized mostly by endothelial cells in response to stimulation by several cytokines.
- Urodilatin: produced by alternative processing of preproANP within the kidney, this hormone appears to act locally in the kidney and to not circulate in blood.
The naturetic peptides bind to three receptors referred to as NPR-A, NPR-B and NPR-C. The A and B receptors are guanylyl cyclases, binding to which increases intracellular concentration of cyclic GMP as a second messenger. The C receptor is a clearance receptor that aids in removing ANP from the cirulation, acting in concert with a number of peptidases that destroy ANP. The plasma halflife of ANP is only 2-4 minutes.
Physiological Effects of Atrial Naturetic Hormone
ANP activity was first demonstrated through a classical endocrine experiment: extracts of atria injected into rats was found to induce increased production of urine (diuresis) and excretion of sodium (naturesis), indicating a major effect on the kidney.
The most potent stimulus for release of ANP is atrial stretch, the consequence of abnormally high circulating blood volume. The desired physiologic response to normalize this condition is to enhance elimination water and sodium in urine. This effect is promoted by two dual activities of ANP within the kidney:
- ANP stimulates vasodilation of the afferent arteriole of glomerulus: this results in increased renal blood flow and an increase in glomerular filtration rate. Increased glomerular filtration, coupled with inhibition of reabsorption, results in increases in excretion of water and urine volume - diuresis! It seems that in most cases, ANP also induced vasoconstriction in the efferent arteriole, which also increases glomerular filtrate volume.
- ANP acts on several segments of the nephron - most notably the inner medullary collecting duct - to reduce reabsorption of sodium. If sodium reabsorption is inhibited, the sodium in filtrate is excreted in urine - naturesis! ANP also inhibits Na+/H+ exchange in the proximal tubule and Na+/Cl- reabsorption in the distal tubule, both of which enhance sodium excretion.
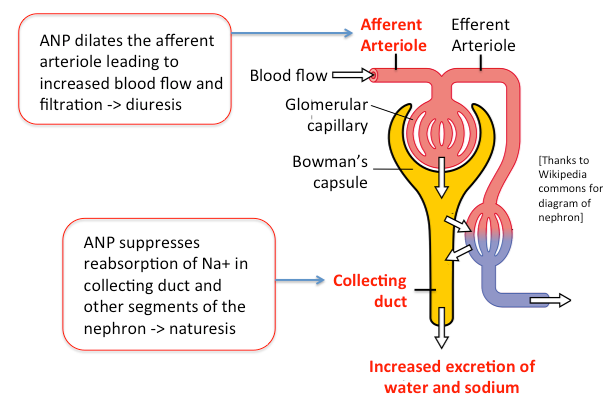
The dual effects of diuresis and naturesis has the effect of reducing effective circulating blood volume and hence blood pressure. With reduction in blood volume, atrial stretch diminishes, thereby removing the stimulus for ANP section.
A number of other effects of ANP enhance its ability to act as an anti-hypertensive hormone. For example, it inhibits release of renin, thereby suppressing generation of angiotensin II, a potent vasoconstrictor.
In addition to it's effect on blood pressure and volume, ANP appears to contribute to several other physiologic and pathologic processes, including inhibiting cardiac hypertrophy in the face of heart failure, promoting cardiomyocyte viability, and modulating a number of immune events.
Disease States
Mice engineered to be incapable of secreting ANP develop chronic hypertension. ANP deficiencies have not been identified in humans, although some types of cardiac surgery can lead to at least temporary deficiency.
ANP and espectially BNP have proven valuable as biomakers of heart failure and are used in monitoring the effect of therapies in such patients.
References and Reviews
- De Vito P. Atrial natriuretic peptide: An old hormone or a new cytokine? Peptides 2014; 58:108-116.
- Kerkel R, Ulvila J, Magga J. Natriuretic peptides in the regulation of cardiovascular physiology and metabolic events. J Am Heart Assoc 2015; 4:e002423 doi: 10.1161/JAHA.115.002423.
- Magnussen C, Blankenberg S. Biomarkers for heart failure: small molecules with high clinical relevance.J Intern Med 2018; 283:530-543.
Updated 2018. Send comments to Richard.Bowen@colostate.edu
