VIVO Pathophysiology
Food Chemistry
The diet of any animal contains hundreds if not thousands of different molecules, but the bulk of the ingested nutrients are in the form of huge macromolecules that cannot be absorbed into blood without first being reduced to much simpler and smaller forms - even table sugar (sucrose) cannot be absorbed without first being enzymatically ripped apart into glucose and fructose. The most important enzymatic reaction in digestion of foodstuffs is hydrolysis - the breaking of a chemical bond by the addition of a water molecule.
Proteins
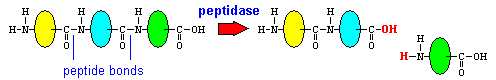
 Proteins are polymers of amino acids linked together by peptide bonds. Chain length varies tremendously and many dietary proteins have been modified after translation by addition of carbohydrate (glycoproteins) or lipid (lipoprotein) moieties. These modifications will be almost totally ignored in this text. Very short proteins, typically 3 to 10 amino acids in length, are called peptides.
Proteins are polymers of amino acids linked together by peptide bonds. Chain length varies tremendously and many dietary proteins have been modified after translation by addition of carbohydrate (glycoproteins) or lipid (lipoprotein) moieties. These modifications will be almost totally ignored in this text. Very short proteins, typically 3 to 10 amino acids in length, are called peptides.
Although very small peptides can be absorbed to a limited degree, for all intents and purposes, proteins must be reduced to single amino acids before they can be absorbed. Enzymes that hydrolyze peptide bonds and reduce proteins or peptides to amino acids are called proteases or peptidases.
Lipids
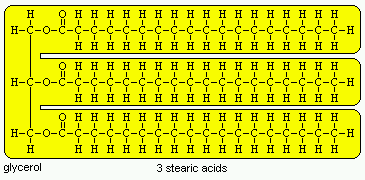
Fatty acids are present in only small amounts in animal and plant tissues, but are the building blocks of many important complex lipids. True fatty acids possess a long hydrocarbon chain terminating in a carboxyl group. Nearly all fatty acids have an even number of carbons and have chains between 14 and 22 carbons in length. The principle differences among the many fatty acids are the length of the chain (usually 16 or 18 carbons) and the positions of unsaturated or double bonds. For example, stearic acid (pictured below) has 18 carbons and is saturated.

The so-called "short-chain" or volatile fatty acids are 2 to 4-carbon molecules of great importance in intermediary metabolism and as the mainstay of ruminant nutrition. They are represented by acetic, butyric and proprionic acids.
 The most abundant storage form of fat in animals and plants, and hence the most important dietary lipid, is neutral fat or triglyceride. A molecule of triglyceride is composed of a molecule of glycerol in which each of the three carbons is linked through an ester bond to a fatty acid. Triglycerides cannot be efficiently absorbed, and are enzymatically digested by pancreatic lipase into a 2-monoglyceride and two free fatty acids, all of which can be absorbed. Other lipases hydrolyse a triglyceride into glycerol and three fatty acids.
The most abundant storage form of fat in animals and plants, and hence the most important dietary lipid, is neutral fat or triglyceride. A molecule of triglyceride is composed of a molecule of glycerol in which each of the three carbons is linked through an ester bond to a fatty acid. Triglycerides cannot be efficiently absorbed, and are enzymatically digested by pancreatic lipase into a 2-monoglyceride and two free fatty acids, all of which can be absorbed. Other lipases hydrolyse a triglyceride into glycerol and three fatty acids.
Carbohydrates
 The diversity of dietary carbohydrates necessitates discussion of several classes of these molecules, ranging from simple sugars to huge, branched polymers.
The diversity of dietary carbohydrates necessitates discussion of several classes of these molecules, ranging from simple sugars to huge, branched polymers.
Monosaccharides or simple sugars are either hexoses (6-carbon) like glucose, galactose and fructose, or pentoses (5-carbon) like ribose. These are the breakdown products of more complex carbohydrates and can be efficiently absorbed across the wall of the digestive tube and transported into blood.
Disaccharides are simply two monosaccharides linked together by a glycosidic bond. The disaccharides most important in nutrition and digestion are:
- lactose or "milk sugar": glucose + galactose
- sucrose or "table sugar": glucose + fructose
- maltose: glucose + glucose
Oligosaccharides, which include disaccharides, are relatively short chains of monosaccharides which typically are intermediates in the breakdown of polysaccharides to monosaccharides.
Polysaccharides are the most abundant dietary carbohydrate for all except very young animals. You should be familiar with three important polysaccharides, each of which is a large polymer of glucose:
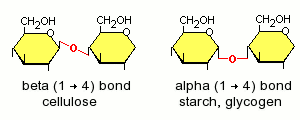
- Starch is a major plant storage form of glucose. It occurs in two forms: alpha-amylose, in which the glucoses are linked together in straight chains, and amylopectin, in which the glucose chains are highly branched. Except for the branch points of amylopectin, the glucose monomers in starch are linked via alpha(1-4) glycosidic bonds, which, in the digestive tract of mammals, are hydrolyzed by amylases.
- Cellulose is the other major plant carbohydrate. It is the major constituent of plant cell walls, and more than half of the organic carbon on earth is found in cellulose. Cellulose is composed on unbranched, linear chains of D-glucose molecules, linked to one another by beta(1-4) glycosidic bonds, which no vertebrate has the capacity to enzymatically digest. Herbivores subsist largely on cellulose, not because they can digest it themselves, but because their digestive tracts teem with microbes that produce cellulases that hydrolyze cellulose.
- Glycogen is the third large polymer of glucose and is the major animal storage carbohydrate. Like starch, the glucose molecules in glycogen are linked together by alpha(1-4) glycosidic bonds.
Advanced and Supplemental Topics
Send comments to Richard.Bowen@colostate.edu

